The native populations of Bolivia’s highlands are a testament to human resilience. For millennia, they have thrived over two miles above sea level, an environment where the oxygen is roughly 35 percent scarcer than at sea level. These conditions are among the most extreme ever faced by humans. It’s well-documented that these Andean inhabitants have genetically adapted to their high-altitude environment. Recently, scientists have discovered another fascinating genetic adaptation that occurred after their ancestors first arrived in the highlands of South America about 10,000 years ago.
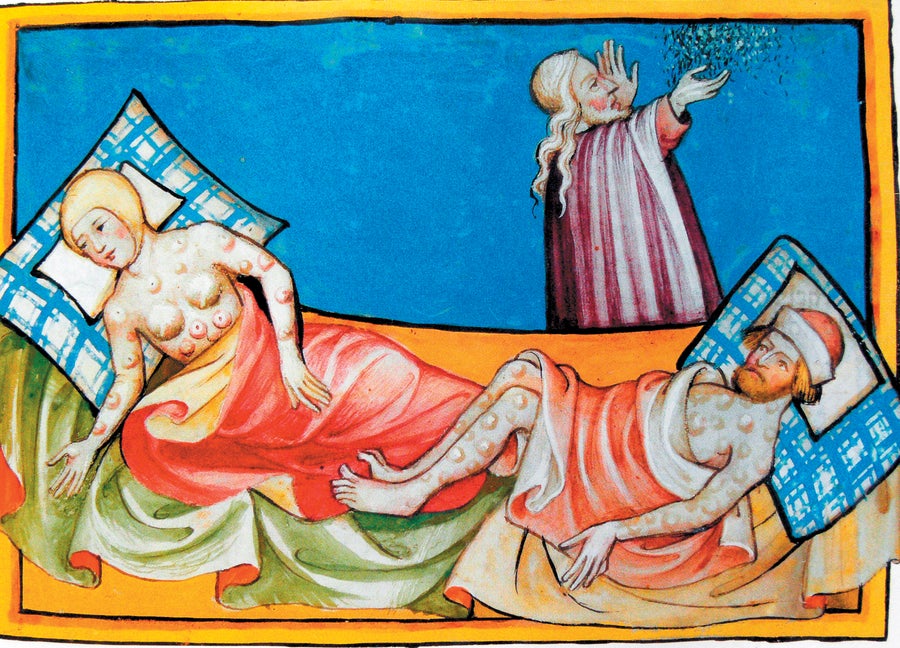
In the Andes, the volcanic terrain naturally contains high levels of arsenic which seeps into the local water supply. The health risks of inorganic arsenic are well-documented, including increased risks of cancer, skin lesions, heart disease, diabetes, and high infant mortality rates in various global populations. However, the biochemistry of the Andean peoples has evolved to efficiently process this toxic element. In Bolivia, as well as in parts of Argentina and Chile, populations have developed genetic variations around the AS3MT gene, which produces enzymes that detoxify arsenic in the liver. This adaptation is a prime example of natural selection, where genetic traits that enhance survival and reproduction become more common over generations. In the Uru, Aymara, and Quechua communities of the Bolivian Altiplano, rare DNA sequences found globally are now common, highlighting a significant case of relatively recent human evolution that challenges long-held views on the subject.
For much of the 21st century, evolutionary biologists have believed that human evolution has been slow in recent millennia compared to earlier periods. The earliest known human ancestors appeared in Africa around six to seven million years ago and exhibited many ape-like features. Our species, Homo sapiens, emerged in Africa a few hundred thousand years ago and started migrating around the globe about 60,000 years ago. By then, our physical traits had largely stabilized, showing only minor variations globally. After millions of years of dramatic evolution from four-legged, small-brained ancestors to large-brained bipeds, it seemed that biological evolution had nearly halted as H. sapiens developed agriculture, established civilizations, and dramatically altered the planet.
Supporting Science Journalism
If you find this content engaging, please consider supporting our award-winning journalism by subscribing. Your subscription helps sustain future reporting on important and impactful stories shaping our world.
Initial DNA studies on modern humans showed few fixed differences—genetic variations unique to certain populations—which seemed to support the idea of evolutionary stasis. Many scientists came to believe that the recent chapters of human history were marked more by cultural than biological changes, such as developing more reliable food sources rather than altering our bodies’ systems.
However, advancements in sequencing both ancient and modern DNA have provided new insights into how our genetic material has evolved over time, revealing numerous instances of natural selection in just the past few thousand years as humans spread into diverse environments with new challenges like unfamiliar foods, diseases, and toxins. Karin Broberg from Sweden’s Karolinska Institute, an expert in genetics related to environmental toxins, notes, “It shows the flexibility of the human genome. We’ve adapted to live in extreme conditions worldwide, much like rats or cockroaches.” This research underscores that cultural adaptation was not the sole driver of our species’ success in colonizing the planet; biological evolution continued to play a critical role.
Understanding these evolutionary developments requires basic knowledge of DNA structure and variation among individuals and populations. The human genome consists of about three billion nucleotide base pairs, which form the building blocks of our genetic code. Today’s human DNA is remarkably similar across the globe; we differ by only about 0.1 percent of our genome. Variations at any specific position in the genome are known as single nucleotide polymorphisms, or SNPs (pronounced “snips”). A genetic variant, which can be a single position or thousands of linked positions that differ between individuals, is called an allele. Generally, human populations share a vast majority of genetic variations and evolutionary history.
New research suggests that recent human history may have involved more dynamic evolution than previously thought.
In classical Darwinian terms, natural selection is often seen as a “hard sweep,” where a beneficial mutation allows certain individuals to live longer or reproduce more, eventually leading to that genetic variant becoming predominant in the population. Early 2000s research began identifying signs of hard sweeps in contemporary genomes, with clear cases often linked to unique environmental challenges. For instance, a mutation approximately 42,000 years ago altered a protein on red blood cells in Africans, enhancing their resistance to malaria. In the Tibetan Highlands, selective sweeps favored genes that improved survival in low-oxygen conditions, although populations in different high-altitude regions like the Himalayas, Andes, and Ethiopian highlands developed distinct genetic adaptations to similar challenges.
Some of the most well-documented selective sweeps occurred in Western Eurasia and were associated with changes in diet, skin pigmentation, and immunity. These sweeps were often tied to major shifts brought on by the advent of agriculture. For example, around 8,500 years ago, early farmers began spreading a genetic variant that allowed them to convert plant-based short-chain fatty acids into essential long-chain polyunsaturated fatty acids, which are crucial for cell membranes, especially in the brain. While hunter-gatherers obtained these nutrients directly from meat and seafood, the new genetic change enabled agriculturalists to derive them from plants. Initially rare, this variant now appears in about 60 percent of Europeans.
The Uru people of the Bolivian Altiplano have a gene variant that helps them metabolize the toxic arsenic found in their drinking water.
Gaston Zilberman
Similarly, as dairy farming became more prevalent, so did a gene variant that allowed people to digest milk into adulthood. When Stonehenge was erected around 5,000 years ago, almost no Europeans had the genes necessary to digest milk after infancy. Like most mammals—and most human populations—our bodies typically stop producing the milk-digesting enzyme lactase after weaning. However, around 4,500 years ago, a gene that kept lactase active in adults began to spread throughout Europe and South Asia. Another series of selective sweeps starting around 8,000 years ago gave Eurasians their distinctive light skin. These changes reduced their production of the dark skin pigment melanin, which likely helped early farmers synthesize vitamin D under sun-scarce conditions.
These well-known examples of hard sweeps were striking because they appeared relatively rare. However, over the past two decades, studies have shown that human populations today have relatively few fixed differences, leading many researchers to conclude that selective sweeps were only a minor part of the genetic changes our species has experienced over the last several thousand years. Most changes, they suggested, arose not from natural selection but from gene flow (as populations mingled due to migration) and genetic drift (random changes in gene frequency).
Yet reconstructing the past from the genomes of modern-day people is a complex task because evolution often obscures its own traces. Early studies based on modern DNA could only detect long-lasting evolutionary impacts. Episodes of natural selection are sometimes temporary, and their signs can disappear from our genomes when selective pressures diminish or populations interbreed. Now, however, ancient DNA is helping researchers uncover instances of past selection that have since been masked.
The sequencing of the first ancient human genome in 2010 marked a turning point. Since then, the number of ancient genomes studied has grown to more than 10,000. This expanding dataset allows for more precise analysis of how the three billion positions in the genome have shifted over recent millennia across different global populations. One 2024 study of ancient DNA traced genetic shifts in Europe amid significant migrations and the transition to farming and pastoralism. Researchers examined over 1,600 ancient genomes from as far back as 11,000 years ago up through the Middle Ages, comparing them with more than 400,000 modern genomes from the UK Biobank. They found no evidence of selection when analyzing the modern genomes alone. However, when they included the ancient genomes, they identified 11 instances of selection. When they further divided these ancient genomes according to ancestral lineages, they discovered 21 instances. This illustrates that to fully understand the scope of natural selection in history, one must examine local populations within specific time frames.
Modern Europeans descend from three main ancestral groups: hunter-gatherers who settled the continent around 40,000 years ago, early farmers from Anatolia who arrived about 8,500 years ago, and pastoralists from the Pontic-Caspian steppe who entered around 5,000 years ago. In 2022, a team led by Yassine Souilmi from the University of Adelaide’s Australian Center for Ancient DNA analyzed 1,162 ancient DNA samples from these groups. They captured snapshots of their genetics before and after these groups mixed, searching the genomes for regions with unusually low or high-frequency alleles, indicators of ancient selective sweeps. They found 57 hard sweeps over the past 50,000 years linked to traits such as fat storage, metabolism, skin physiology, immunity, and neural function—changes thought to be adaptations to colder climates. None of these adaptations were found in a comparative population of sub-Saharan Africans, suggesting they originated after our species spread from its African cradle to other parts of the world.
One notable discovery was a hard sweep on a region of chromosome 6 known as the major histocompatibility complex class III, or MHC III, in ancient Anatolians. This cluster of genes, which play a critical role in immunity, typically shows high genetic diversity to fend off a variety of threats. However, the researchers found the opposite—a significant reduction in genetic diversity in this region. This suggested that these early farmers had faced a severe disease outbreak that eliminated much of the genetic diversity usually favored in that area. “The population had been exposed to something so severe that it wiped out all the diversity that is generally favored in that region,” Souilmi explains. “It was one of the strongest, if not the strongest, adaptation signals we have ever seen in humans.”

Cameron Aldridge combines a scientific mind with a knack for storytelling. Passionate about discoveries and breakthroughs, Cameron unravels complex scientific advancements in a way that’s both informative and entertaining.
